
Later that decade, Dmitri Mendeleev, a Russian chemist, organized all the known elements according to similar properties. In 1864, Julius Lothar Meyer, a German chemist, organized the elements by atomic mass and grouped them according to their chemical properties. Likewise, lithium, sodium, and potassium react with other elements (such as oxygen) to make similar compounds. For example, chlorine, bromine, and iodine react with other elements (such as sodium) to make similar compounds. In the 19th century, many previously unknown elements were discovered, and scientists noted that certain sets of elements had similar chemical properties. Greenhouse gases and their relative effects.Oxides of nitrogen and sulfur: sources of pollution (natural, domestic, industrial, internal combustion engine).Acid rain and its effects on the environment.Limitations of the pH scale - usefulness confined to dilute aqueous solutions. 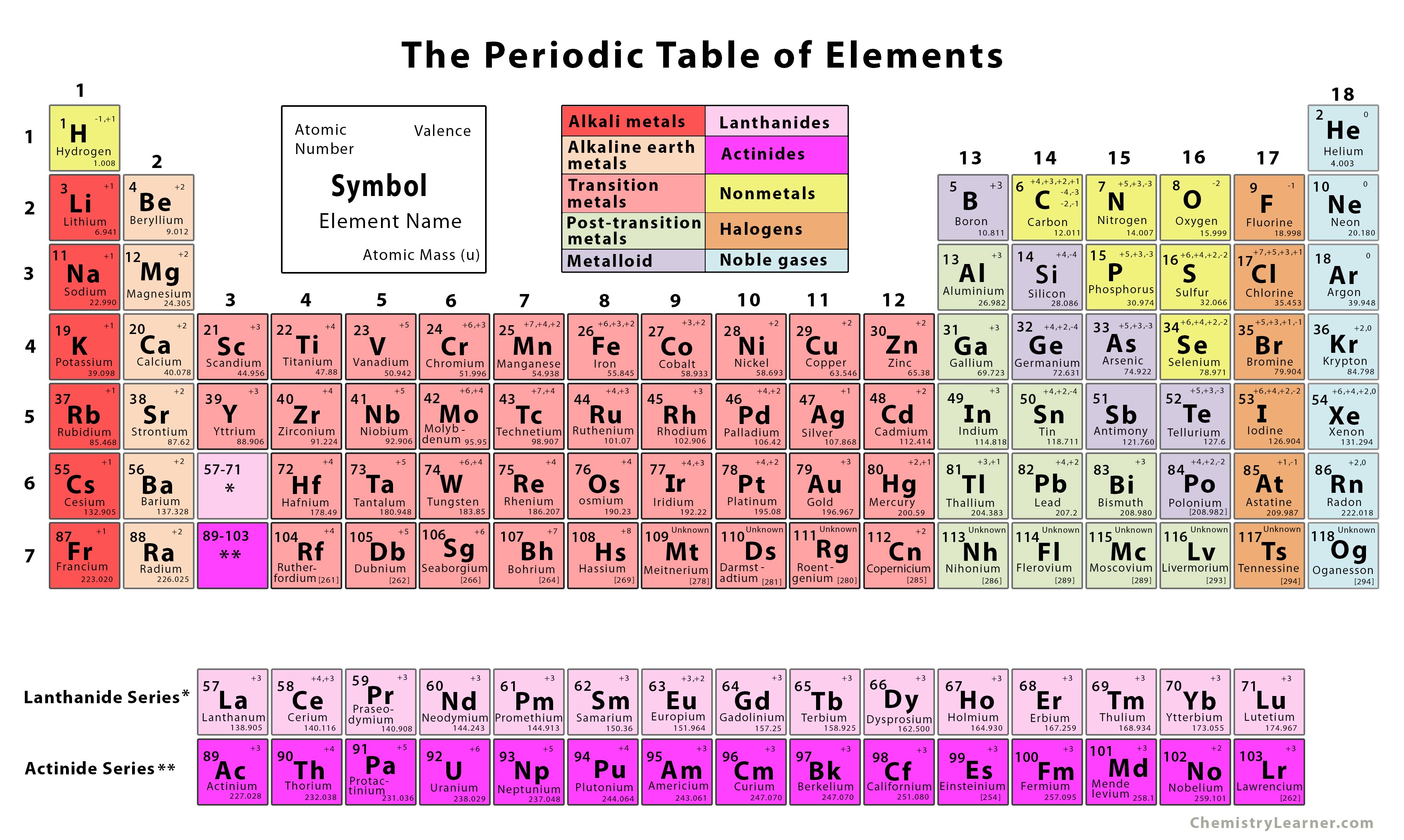
Use of universal indicator paper or solution.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
